Plasma COVID therapy pioneers seek more blood donors
By Lim Jeong-yeoPublished : July 12, 2020 - 17:56
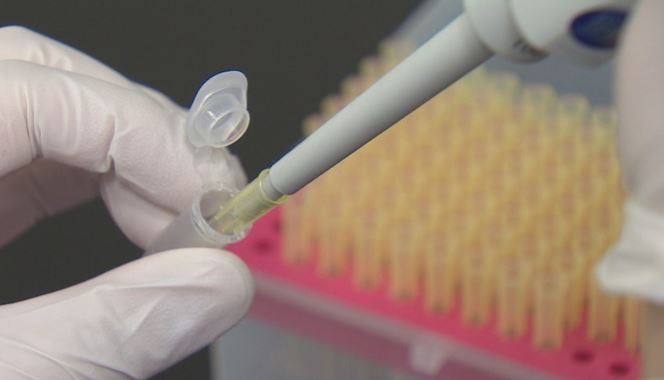
Expectations are growing that South Korea will have plasma-derived COVID-19 therapy by the end of this year, but with the lack of blood donors, the deadline appears unrealistic.
GC Pharma, which is leading the research and development of Korea’s plasma-derived therapy for COVID-19, aims to make its GC5131A pipeline commercially available this year.
The drug will not eradicate COVID-19, but will lower the death toll among critically ill patients, a GC Pharma official told The Korea Herald.
GC Pharma CEO Huh Eun-chul had promised in May that the plasma-derived treatment would be free for all COVID-19 patients in Korea.
However, out of some 12,000 recovered COVID-19 patients who could donate blood for the cause, just 361 have so far shown interest and only 171 had volunteered as of Saturday.
To produce enough for 60 patients, at least 100 people must donate plasma. And since not everyone has identical amounts of antibodies in their plasma, even that might not be enough.
Even if more people are willing to give blood, only four hospitals in the country are willing to assist the plasma donations. Only one hospital in Seoul and three in Daegu currently provide the service, and each hospital can process only five donations per day.
These are Korea University Ansan Hospital in Seoul, and Kyungpook National University Hospital, Keimyung University Dongsan Medical Center and Daegu Fatima Hospital in Daegu.
A GC Pharma official said the company is encouraging more hospitals to take part. However, many are reluctant due to the burden of a heavier workload and the risk of re-inviting recovered COVID-19 patients to the hospital.
Instead, it is possible that the Korean Red Cross may step in to help by offering the use of its blood donation buses, the GC Pharma official said.
Despite the concerns, GC Pharma officials maintain that the plasma drive is ongoing and the clinical trials will pan out according to plan. The company has enough plasma to make drug samples for the clinical trials, and they will be manufactured within this week.
Plasma-derived therapies use naturally formed antibodies found in the recovered COVID-19 patients’ blood and therefore take less time to develop than other biologic drugs. They present no risks to human health.
GC Pharma said it plans to begin clinical phase 2 or 3 trials of its GC5131A plasma therapy pipeline within July, skipping the phase 1 trials in a fast-track development decision by the drug authorities.
GC Pharma is sourcing the plasma through the government and is jointly developing the therapy with the Korea National Institute of Health.
What may be a saving bell for the plasma-derived therapy’s mass production may surprisingly be the Sincheonji Church followers -- believed to be the reason for the initial spread of the disease in the nation.
Some 4,000 Sincheonji believers have said they are willing to donate plasma. In an event that is separate from the first round of clinical trials, starting Monday, 500 followers of the Sincheonji Church of Jesus will give plasma to the KCDC.
By Lim Jeong-yeo (kaylalim@heraldcorp.com)
GC Pharma, which is leading the research and development of Korea’s plasma-derived therapy for COVID-19, aims to make its GC5131A pipeline commercially available this year.
The drug will not eradicate COVID-19, but will lower the death toll among critically ill patients, a GC Pharma official told The Korea Herald.
GC Pharma CEO Huh Eun-chul had promised in May that the plasma-derived treatment would be free for all COVID-19 patients in Korea.
However, out of some 12,000 recovered COVID-19 patients who could donate blood for the cause, just 361 have so far shown interest and only 171 had volunteered as of Saturday.
To produce enough for 60 patients, at least 100 people must donate plasma. And since not everyone has identical amounts of antibodies in their plasma, even that might not be enough.
Even if more people are willing to give blood, only four hospitals in the country are willing to assist the plasma donations. Only one hospital in Seoul and three in Daegu currently provide the service, and each hospital can process only five donations per day.
These are Korea University Ansan Hospital in Seoul, and Kyungpook National University Hospital, Keimyung University Dongsan Medical Center and Daegu Fatima Hospital in Daegu.
A GC Pharma official said the company is encouraging more hospitals to take part. However, many are reluctant due to the burden of a heavier workload and the risk of re-inviting recovered COVID-19 patients to the hospital.
Instead, it is possible that the Korean Red Cross may step in to help by offering the use of its blood donation buses, the GC Pharma official said.
Despite the concerns, GC Pharma officials maintain that the plasma drive is ongoing and the clinical trials will pan out according to plan. The company has enough plasma to make drug samples for the clinical trials, and they will be manufactured within this week.
Plasma-derived therapies use naturally formed antibodies found in the recovered COVID-19 patients’ blood and therefore take less time to develop than other biologic drugs. They present no risks to human health.
GC Pharma said it plans to begin clinical phase 2 or 3 trials of its GC5131A plasma therapy pipeline within July, skipping the phase 1 trials in a fast-track development decision by the drug authorities.
GC Pharma is sourcing the plasma through the government and is jointly developing the therapy with the Korea National Institute of Health.
What may be a saving bell for the plasma-derived therapy’s mass production may surprisingly be the Sincheonji Church followers -- believed to be the reason for the initial spread of the disease in the nation.
Some 4,000 Sincheonji believers have said they are willing to donate plasma. In an event that is separate from the first round of clinical trials, starting Monday, 500 followers of the Sincheonji Church of Jesus will give plasma to the KCDC.
By Lim Jeong-yeo (kaylalim@heraldcorp.com)



![[Exclusive] Korean military set to ban iPhones over 'security' concerns](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/23/20240423050599_0.jpg&u=20240423183955)




![[Pressure points] Leggings in public: Fashion statement or social faux pas?](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/23/20240423050669_0.jpg&u=)
![[Herald Interview] 'Amid aging population, Korea to invite more young professionals from overseas'](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/24/20240424050844_0.jpg&u=20240424200058)









