Search Results
You searched for "Merck" ( 316 results )
-
Merck showcases automotive solutions at Frankfurt motor show
The US science and technology firm Merck said Monday it is participating for the first time in the International Motor Show that began last week, presenting its solutions for the automotive industry that enhance functionality and aesthetics in automotive engineering.The International Motor Show began Thursday in Frankfurt, Germany, and will run for 10 days. Merck said its presentations for the automotive industry include materials for lighting systems, sunroofs, free-form displays and smart ante
Industry Sept. 19, 2017

-
SK Chemicals, GSK awaiting commercial sales of shingles vaccine
SK Chemicals Co., a South Korean pharmaceutical and chemical goods manufacturer, and multinational drug firm GlaxoSmithKline Plc. are awaiting final approval of their respective herpes zoster vaccines, market watchers said Tuesday.Approval is forecast to shake up the market in a big way, setting up a showdown with the US pharmaceutical giant Merck & Co's Zostavax, they said.Zostavax is the only vaccine currently approved for use to treat shingles, which typically shows up as a painful rash that
Industry Sept. 19, 2017

-
Samsung Bioepis on track to launching first Herceptin biosimilar in Europe
Samsung Bioepis has become the first company to secure Europe’s backing of its copy of Roche’s blockbuster breast cancer therapy Herceptin, marking a milestone for the South Korean drugmaker, which specializes in making cheaper copies of complex biologic drugs known as biosimilars. The European Medicines Agency’s Committee for Medicinal Products for Human Use on Friday recommended the approval of Samsung’s Herceptin-referencing biosimilar Ontruzant. It is the first biosimilar copy of Herceptin t
Industry Sept. 18, 2017

-
Merck features textile facade based on OPV, OLED tech at Seoul Biennale
German display materials maker Merck on Friday unveiled a new textile facade concept combining organic photovoltaics and organic light-emitting diodes technologies at the Seoul Biennale of Architecture and Urbanism. The facade concept was prepared by Merck in partnership with OledWorks, OPVIUS and South Korean chemicals company Kolon. German architects Nikolaus Hirsch and Michel Muller as well as New York-based Thai artist Rirkrit Tiravanija designed the work as part of their ongoing project cal
Technology Sept. 1, 2017

-
Korea approves LG Chem’s combination drug for diabetes, dyslipidemia
LG Chem said Wednesday that South Korea’s Ministry of Food and Drug Safety had approved Zemiro, a combination drug that simultaneously treats diabetes and dyslipidemia, a condition in which an abnormal amount of lipids exist in the blood. Developed by LG Chem’s biopharmaceuticals unit, Zemiro is a new combination tablet that merges LG’s diabetes drug Zemiglo -- a Diepeptidyl peptidase-4 inhibitor -- with the lipid reduction drug ingredient rosuvastatin. LG Chem`s production plant in Osong, Nort
Industry Aug. 2, 2017

-
Samsung Bioepis, Merck & Co. launch Remicade biosimilar in US
Samsung Bioepis said Tuesday that it has begun selling a cheaper biosimilar drug referencing Johnson & Johnson’s Remicade in the US via its partner Merck & Co., marking the second Remicade copy to hit the US market. The launch of Samsung’s Remicade biosimilar, named Renflexis, comes three months after its official approval by the US Food and Drug Administration in April.Renflexis was developed by Songo-based Samsung Bioepis and is being commercialized in the US by its global sales marketing part
Industry July 25, 2017

-
Samsung Bioepis biosimilar wins tentative approval in US
Samsung Bioepis Co. diabetes treatment biosimilar received tentative approval from the US Food and Drug Administration, industry sources said Sunday. (Yonhap)The Lusduna Nexvue developed in cooperation with multinational drug company Merck Sharp and Dohme Corp. got the green light because it satisfied all regulatory rules for follow-on biologics of clinical and nonclinical safety, local here observers said.A biosimilar is a biologic medical product which is almost an identical copy of an origina
Industry July 23, 2017

-
Ransomware cyberattack goes global
A new cyberattack similar to WannaCry reached parts of Asia after hitting businesses, port operators and government systems in Europe, US and South America.A terminal operated by A.P. Moller-Maersk at the Jawaharlal Nehru Port Trust, a facility near Mumbai which is India’s biggest container port, was unable to load or unload because of the attack. With the Gateway Terminal India facility unable to identify which shipment belongs to whom, the port is clearing cargo manually, Chairman Anil Diggika
World News June 28, 2017
-
[Newsmaker] New cyberattack wallops Europe, spreads slowly in US
A new and highly virulent outbreak of data-scrambling software -- apparently sown in Ukraine -- caused disruption across the world Tuesday. Following a similar attack in May, the fresh cyber assault paralyzed some hospitals, government offices and major multinational corporations in a dramatic demonstration of how easily malicious programs can bring daily life to a halt. Ukraine and Russia appeared hardest hit by the new strain of ransomware -- malicious software that locks up computer files wit
World News June 28, 2017
![[Newsmaker] New cyberattack wallops Europe, spreads slowly in US](//res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2017/06/28/20170628000994_0.jpg&u=20170628164150)
-
US Supreme Court decision speeds up entry of biosimilars to market
The US Supreme Court on Tuesday ruled that biosimilar developers do not have to wait six months after regulatory approval to bring their products to market, marking a boon for companies developing the new class of cheaper, near-replicas of expensive biologic drugs for the US. The landmark decision is expected to benefit biosimilar developers around the world, including South Korea’s Celltrion as well as Samsung Bioepis, which are planning to commercialize their products in the US in the future.
Industry June 14, 2017

-
Korean pharmaceutical companies to release important drugs this year
South Korean pharmaceutical companies are poised to release a series of important drugs in the second half of this year that could change the existing market, industry sources said Friday. Kolon Life Science's Invossa, a gene drug for osteoarthritis, will receive an approval from the Ministry of Food and Drug Safety later this month at the earliest, according to sources. This would make it the world's first medicine for the degenerative joint disease. This file photo taken on Aug. 23, 2016, sho
Industry June 9, 2017

-
[Herald Interview] We think in generations, not in quarters: Merck chairman
It all started from pure scientific curiosity when Merck decided to develop liquid crystal in 1903. The organic substance, however, remained in the research and development territory for more than 50 years until it started to be applied to wrist watches, calculators and computers -- products that ushered in a new era of technology in the 20th century. Therefore, the German company based in Darmstadt, did the same with the organic light-emitting diode, known as a future technology for the 21st ce
Industry May 23, 2017
![[Herald Interview] We think in generations, not in quarters: Merck chairman](//res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2017/05/23/20170523001019_0.jpg&u=20170523173428)
-
Sales of Celltrion’s Remsima affect Q1 earnings of Pfizer, MSD
The first-quarter earnings of Pfizer and US-based Merck & Co. appear to have been affected by sales of Celltrion’s Remsima, a biosimilar copy of Johnson & Johnson’s Remicade. Pfizer, which markets and sells Celltrion’s Remicade biosimilar in the US and other global markets, said in its earnings report for the January-March period Wednesday that worldwide sales of its biosimilars in the quarter came to $105 million. Of this amount, global sales of Remsima reached $78 million. Remsima is Celltrion
Industry May 4, 2017

-
Samsung Bioepis’ Remicade biosimilar wins US FDA approval
South Korea’s Samsung Bioepis won the US Food and Drug Administration’s approval for its biosimilar copy of Johnson & Johnson’s Remicade on Saturday, becoming a new challenger to the sales of the pharmaceutical giant’s blockbuster autoimmune disease treatment in the US. The US FDA has granted approval to Samsung’s Renflexis, a biosimilar referencing Remicade (infliximab). Akin to generics of chemical drugs, biosimilars refer to cheaper, near-replicas of live cell biologic drugs whose patents hav
Industry April 23, 2017

-
Johnson & Johnson Innovation eyes Korean tie-ups
The chief of Johnson & Johnson Innovation, the strategic venture arm of the US health care giant leading the firm’s open innovation push, is to hold preliminary meetings with a number of South Korean biotech and medical tech firms this week.Robert Urban, the global head of Johnson & Johnson Innovation, told The Korea Herald Tuesday that his team would discuss Korea’s health care ecosystem and J&J’s innovation platform with four to five local biopharmaceutical companies and investor groups over t
Industry April 11, 2017

-
Merck generates record sales in 2016
German pharmaceutical company Merck said Monday its sales and earnings rose significantly in 2016, as major strategic advances were made in all three business sectors.“2016 was a successful year for Merck. In health care, two of our compounds are in registration. In our life science business sector, we made rapid progress with the integration of Sigma-Aldrich. We have moved ahead faster and even better than expected with the realization of synergies,” said Merck CEO through the company’s stateme
Industry March 14, 2017
-
Europe approves Lantus biosimilar developed by MSD
The European Medicines Agency has granted official sales approval to a new Lantus biosimilar developed by US-based Merck Sharp & Dohme with partial funding from Samsung Bioepis. The European drug regulator approved MSD’s Lusduna -- a biosimilar drug referencing Lantus (insulin glargine), an insulin-based diabetes treatment originally developed by Sanofi -- on Jan. 4. It is the second Lantus biosimilar to be approved by the EMA after Eli Lilly’s Basaglar, another biosimilar referencing Sanofi’s t
Industry Jan. 25, 2017

-
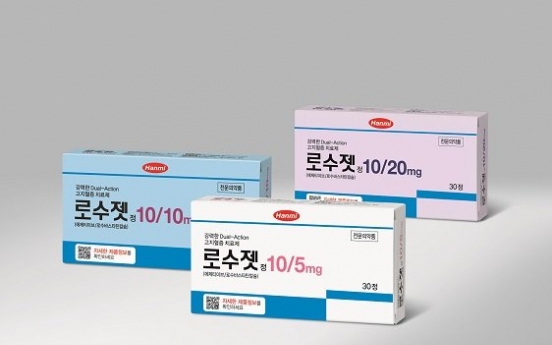
Hanmi to globally launch dual-action hyperlipidemia drug with MSD
South Korea’s Hanmi Pharmaceutical said Wednesday it will launch its dual-action drug for hyperlipidemia, Rosuzet, across global markets in partnership with US pharmaceutical giant Merck Sharp & Dohme. The two have newly entered a licensing partnership under which MSD will take charge of selling Hanmi’s Rosuzet across 23 countries, excluding Korea. Hanmi said it could not specify the given countries due to contract stipulations. Hanmi Pharmaceutical's Rosuzet (Hanmi Pharmaceutical)Rosuzet is a d
Industry Jan. 4, 2017
-
[ECONOMY AT RISK] Hurdles lie ahead for Korea's biotechnology push
The Korea Herald is publishing a series of articles on the alarming state of the country’s economy and the challenges to be addressed. This is the fifth installment. -- Ed.Faced with the continued slowdown of its manufacturing sector, South Korea is eyeing new growth in the field of biotechnology, a promising industry expected for a sharp growth in the years ahead. Biotechnology broadly refers to the use of genetic engineering and molecular biology to modify living cells to produce novel substan
Industry Nov. 13, 2016
![[ECONOMY AT RISK] Hurdles lie ahead for Korea's biotechnology push](//res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2016/11/13/20161113000174_0.jpg&u=20161113161054)
-
美머로우소달리 “주주들 엘리엇 의도 관심없다...삼성 이사회 판단력이 관건”
[코리아헤럴드(더인베스터)=박한나 기자] 삼성그룹 지배구조에 대한 미국계 헤지펀드 엘리엇의 공세가 거세지고 있는 가운데 정작 대부분의 주주들이 관심을 갖는 것은 엘리엇의 의도 자체보다는 삼성 이사회의 판단력과 커뮤니케이션 능력이라고 미국의 저명한 지배구조 전문가가 평했다. (사진=머로우소달리 존 윌콕스 회장)글로벌 1위 지배구조 컨설팅 회사 머로우소달리 (Morrow Sodali)의 존 윌콕스 회장은 10일 더인베스터와의 인터뷰에서 “어떤 행동주의 투자자들은 단기적 이득을 얻기 위해 기업을 곤란에 빠트리는가 하면 또 일부 행동주의 투자자들은 회사의 근본적인 기업지배구조, 자본 배분 등에 깊은 관심을 갖는다. 그들의 의도를 파악하는 것은 온전히 기업의 몫”이라며 “행동주의 투자자들의 제안을 받아들이느냐 아니냐는 기업의 장기적 목표화 비즈니스 전략을 가장 잘 아는 기업자체와 그 이사회가 선별적으로 판단해야 한다”고 말했다.지난 10월 엘리엇은 서한을 통해 삼성전자에 인적분할, 특별배당
한국어판 Nov. 11, 2016

Most Popular
-
1
Tensions heighten ahead of first president-opposition chief meeting

-
2
Seoul to provide housing subsidy to married couples with newborns

-
3
[KH Explains] No more 'Michael' at Kakao Games
![[KH Explains] No more 'Michael' at Kakao Games](//res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/28/20240428050183_0.jpg&u=20240428180321)
-
4
Rapper jailed after public street fight with another rapper

-
5
Woman gets suspended term for injuring boyfriend with knife

-
6
Samsung chief bolsters ties with Germany’s Zeiss

-
7
Nominee for chief of anti-corruption body pledges 'independence, effectiveness'

-
8
NewJeans pops out ‘Bubble Gum’ video amid troubles at agency
-
9
Med schools expect 1,500+ new admission slots next year

-
10
KT launches new mobile plans for foreign residents











![[Newsmaker] New cyberattack wallops Europe, spreads slowly in US](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2017/06/28/20170628000994_0.jpg&u=20170628164150)


![[Herald Interview] We think in generations, not in quarters: Merck chairman](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2017/05/23/20170523001019_0.jpg&u=20170523173428)





![[ECONOMY AT RISK] Hurdles lie ahead for Korea's biotechnology push](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=649&simg=/content/image/2016/11/13/20161113000174_0.jpg&u=20161113161054)



![[KH Explains] No more 'Michael' at Kakao Games](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/28/20240428050183_0.jpg&u=20240428180321)