Moderna nears deal with Korea to provide 40 million doses
US troops receive first vaccines in the country
By Kim ArinPublished : Dec. 29, 2020 - 18:01
South Korea and Moderna are close to reaching a deal for a supply of 40 million doses, the first of which will be delivered no later than summer next year, according to Cheong Wa Dae on Tuesday.
Under the agreement, the US pharmaceutical company will be providing the country 20 million doses more than what was initially announced, with their distribution here a few months faster.
The negotiations took place during a videoconference between President Moon Jae-in and Moderna Chief Executive Officer Stephane Bancel on Monday evening, the presidential office said.
“We hope to finalize the deal before the year is over,” the president was quoted as saying, to which Bancel is said to have responded affirmatively.
The supply deals announced so far, both imminent and finalized, will give Korea access to enough vaccines for 56 million people in total -- more than enough to cover the country’s population. A source familiar with the discussions said more doses were expected to be procured through additional deals with companies such as Novavax.
Last week Korea said it had closed deals with Pfizer and Janssen to acquire a combined 26 million doses of their vaccines, in addition to the 20 million doses of AstraZeneca’s the country had locked in through an advance purchase agreement earlier this month.
With the exception of Janssen’s vaccine, which is a single-dose product, the vaccines are to be administered in two doses several weeks apart.
Korea is also set to acquire vaccines for 10 million people through the COVAX facility, a World Health Organization initiative for fair and equitable vaccine distribution.
The Korea Disease Control and Prevention Agency told a news briefing Tuesday that the country would be able to start vaccinations in February or March. The first batch of the purchased vaccines to arrive here will be AstraZeneca’s.
Front-line health care workers and vulnerable people are first in line to get the vaccines. The agency officials estimate that it will take until November to vaccinate those groups.
Among the companies the country has struck or is set to strike deals with, Pfizer and Moderna are the only two to have won emergency use authorization from the US Food and Drug Safety Administration.
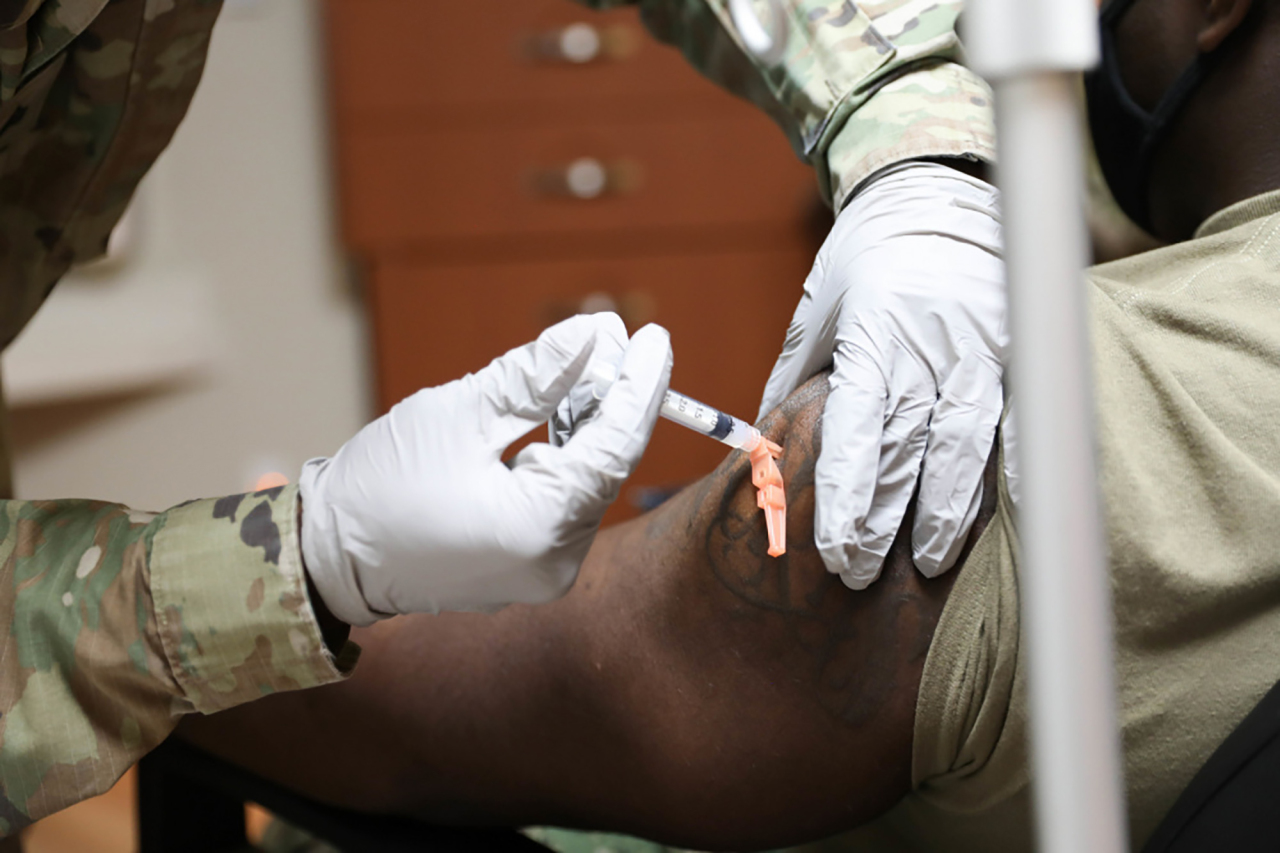
American troops stationed in Korea became the first in the country to receive COVID-19 vaccinations on Tuesday.
United States Forces Korea said in a press release that it has started inoculating military and civilian health care workers, first responders and the command team with the Moderna COVID-19 vaccine across three medical treatment facilities.
Korea is among the initial sites for the US Department of Defense’s phased vaccine distribution plan.
“I strongly encourage all eligible individuals to receive the vaccine,” said USFK Commander Gen. Robert B. Abrams. Under emergency use authorization, the vaccine is voluntary and not mandatory.
Following this initial phase of inoculations, additional shipments of Moderna, and potentially other FDA-authorized vaccines, will arrive at USFK.
On the same day, Korea posted new highs for COVID-19 cases and deaths. There were 1,046 new cases and 40 deaths reported -- the highest numbers in a single day -- according to the national health agency’s situation report.
December is on track to be the worst month yet in Korea’s epidemic. One Korean has died of COVID-19 every two hours so far this month. Nearly half of the total 58,725 cases confirmed to date were reported this month.
A detention center in Songpa, southern Seoul, became the site of the largest cluster of infections tied to a single facility, with 769 cases identified to date. One of the inmates died Sunday, three days after testing positive for the virus.
By Kim Arin (arin@heraldcorp.com)









![[K-pop’s dilemma] Time, profit pressures work against originality](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/05/08/20240508050705_0.jpg&u=20240508171126)









![[Today’s K-pop] Stray Kids to drop new album in July: report](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=642&simg=/content/image/2024/05/09/20240509050659_0.jpg&u=)