Daewoong claims its BTX strain different from Medytox
By Shin Ji-hyePublished : Sept. 5, 2019 - 15:21
South Korea’s Daewoong Pharmaceutical said Thursday it is close to settling a legal dispute with its local rival Medytox, which accused it of stealing trade secrets through a former researcher.
Daewoong’s Nabota, sold as Jeuveau overseas, is a neurotoxin drug to treat forehead wrinkles. The drug, approved by the US Food and Drug Administration in January, was to be commercialized by California-based firm Evolus in the second quarter of this year.
Daewoong’s Nabota, sold as Jeuveau overseas, is a neurotoxin drug to treat forehead wrinkles. The drug, approved by the US Food and Drug Administration in January, was to be commercialized by California-based firm Evolus in the second quarter of this year.
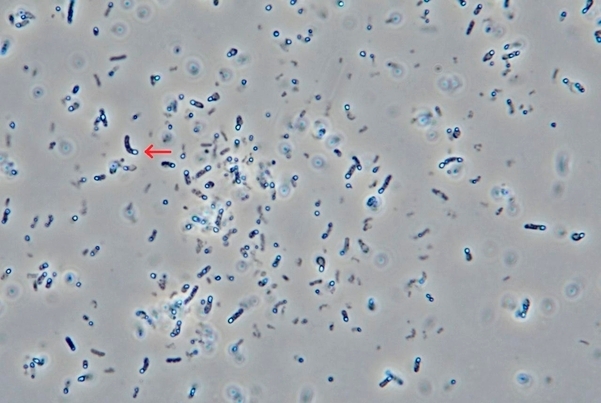
In January, however, Medytox approached the US International Trade Commission claiming Daewoong developed the drug with a stolen botulinum toxin (BTX) strain. It argued Medytox’s former employee had taken the samples and transferred them to Daewoong.
On Thursday, Daewoong released a statement, saying it found evidence that its strains are different from Medytox through an experiment in preparation for the ITC investigation.
“Medytox had claimed in its complaint to the ITC that its strains do not form an endospore in any conditions because they die when exposed to air or deprived of it,” the company said.
“However, we found our strains formed an endospore during an appraisal test, which means there is a difference.”
Medytox did not agree with Daewoong’s claim, saying, “All allegations will be identified through the investigation results to be submitted by the two firms by Sept. 22.”
By Shin Ji-hye (shinjh@heraldcorp.com)






![[Graphic News] Number of coffee franchises in S. Korea rises 13%](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/05/02/20240502050817_0.gif&u=)

![[Robert J. Fouser] AI changes rationale for learning languages](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/05/02/20240502050811_0.jpg&u=)








![[Eye Interview] 'If you live to 100, you might as well be happy,' says 88-year-old bestselling essayist](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=652&simg=/content/image/2024/05/03/20240503050674_0.jpg&u=)
