[Newsmaker] [Feature] Debunking Saxenda myth
No weight-loss shortcuts have lasting effects: experts
By Lim Jeong-yeoPublished : April 28, 2019 - 15:30
In recent months, an unusual buzzword has spread among trendsetters, social media influencers and the general public. Injectable medicine “Saxenda” is being touted across various circles as one of the quickest and most efficient ways to lose weight, by suppressing one’s appetite.
Although a prescription-based medicine, there were soon reports of people buying them in bulk through backdoor dealings and others reselling them.
Saxenda (liraglutide) injections soon soared to the No. 3 spot in Korea’s market for weight control treatments, less than a year after launching in March 2018, according to health data research firm IQVIA.
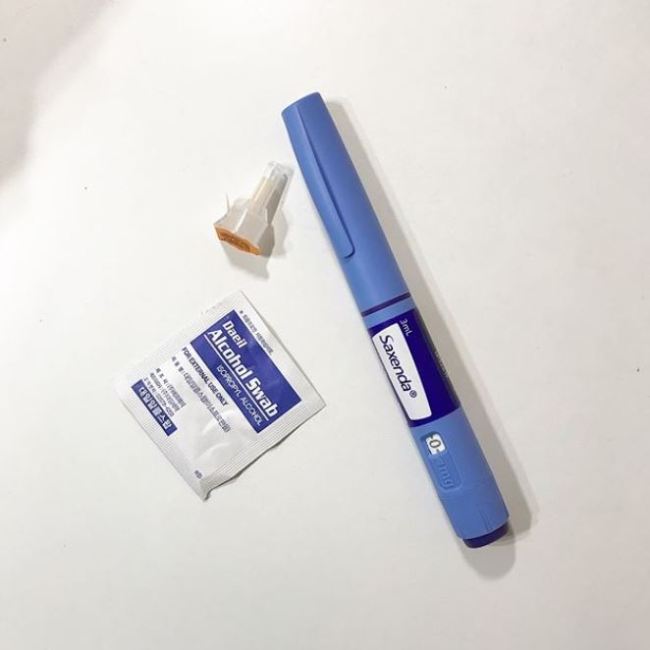
Made by Danish pharma company Novo Nordisk, Saxenda was developed as a diabetes treatment, but following revelations it also showed efficacy for weight loss, it was widely marketed as a means of weight control.
Saxenda’s glucagon-like peptide 1 induces a gastric hormone that gives the feeling of fullness and suppresses the appetite. In a clinical trial for Saxenda carried out on 3,731 obese or prediabetic patients over 56 weeks accompanied with an active lifestyle and balanced diet, 90 percent of patients marked decreased weight. Among them, 63 percent lost over 5 percent of their body weight, 33 percent lost over 10 percent and 14 percent lost more than 15 percent. Just 1 in 10 people showed no response to Saxenda.
But with abnormal hype over the injection treatment, experts have started to underscore that there is no shortcut to weight loss -- only the tried and tested method of exercise and a balanced diet, and that unadvised or excessive use of any medical treatment can come with side effects.
“Saxenda is no use to people who have average body mass index,” said Kim Chung-ha, a professor of Family Medicine at Chung-Ang University Hospital.
The drug should have no strong effects for lightweights. Saxenda is advised by its maker Novo Nordisk and Korea’s Ministry of Food and Drug Safety as a prescription drug for people who are obese, whose body mass index is over 30, or 27 with risk of developing diabetes, high blood pressure or dyslipidemia.
“Social media is often the first source my patients quote when they come looking for Saxenda,” Kim said. “They confuse tackling obesity with beauty procedures. Saxenda should only be used by people with weight control issues.”
Market hype
This reporter, with a classified normal BMI of 20.9, visited a nearby OB-GYN clinic for a consultation on a Saxenda prescription. The female consultant behind the counter assured the reporter, “It’s all about patient satisfaction. You can take it if you want it.” All five female clerks there -- all slim in their tight-fitting uniforms -- were said to have used Saxenda.
The pen-type injection sells for variable prices of 100,000 won ($87) to 140,000 won at hospitals. A purchase of five pens in a pack is common, as observed on social media. Although a doctor’s prescription is required for the Saxenda purchase, this is not the only route people take.
Although a prescription-based medicine, there were soon reports of people buying them in bulk through backdoor dealings and others reselling them.
Saxenda (liraglutide) injections soon soared to the No. 3 spot in Korea’s market for weight control treatments, less than a year after launching in March 2018, according to health data research firm IQVIA.

Made by Danish pharma company Novo Nordisk, Saxenda was developed as a diabetes treatment, but following revelations it also showed efficacy for weight loss, it was widely marketed as a means of weight control.
Saxenda’s glucagon-like peptide 1 induces a gastric hormone that gives the feeling of fullness and suppresses the appetite. In a clinical trial for Saxenda carried out on 3,731 obese or prediabetic patients over 56 weeks accompanied with an active lifestyle and balanced diet, 90 percent of patients marked decreased weight. Among them, 63 percent lost over 5 percent of their body weight, 33 percent lost over 10 percent and 14 percent lost more than 15 percent. Just 1 in 10 people showed no response to Saxenda.
But with abnormal hype over the injection treatment, experts have started to underscore that there is no shortcut to weight loss -- only the tried and tested method of exercise and a balanced diet, and that unadvised or excessive use of any medical treatment can come with side effects.
“Saxenda is no use to people who have average body mass index,” said Kim Chung-ha, a professor of Family Medicine at Chung-Ang University Hospital.
The drug should have no strong effects for lightweights. Saxenda is advised by its maker Novo Nordisk and Korea’s Ministry of Food and Drug Safety as a prescription drug for people who are obese, whose body mass index is over 30, or 27 with risk of developing diabetes, high blood pressure or dyslipidemia.
“Social media is often the first source my patients quote when they come looking for Saxenda,” Kim said. “They confuse tackling obesity with beauty procedures. Saxenda should only be used by people with weight control issues.”
Market hype
This reporter, with a classified normal BMI of 20.9, visited a nearby OB-GYN clinic for a consultation on a Saxenda prescription. The female consultant behind the counter assured the reporter, “It’s all about patient satisfaction. You can take it if you want it.” All five female clerks there -- all slim in their tight-fitting uniforms -- were said to have used Saxenda.
The pen-type injection sells for variable prices of 100,000 won ($87) to 140,000 won at hospitals. A purchase of five pens in a pack is common, as observed on social media. Although a doctor’s prescription is required for the Saxenda purchase, this is not the only route people take.

“A friend of mine got this for me because I was lately depressed for having put on weight,” a woman brazenly boasted on Instagram, her thin wrist holding a Saxenda pen. It’s illegal to sell or trade Saxenda between consumers. The secondhand sales of Saxenda is punishable by a maximum five years in prison or 50 million won in fines, according to the Pharmaceutical Affairs Act.
“It has come to our attention that some people, who bought Saxenda expecting dramatic results instead suffered prenotified side effects of nausea and gastric discomfort and decided to resell the unused pens from the pack through social media or the online secondhand market,” said a Korean Medical Association spokesperson.
Proper use
To prevent illegal trade, the KMA urges patients to always consult a doctor after each Saxenda pen use. The pens each last for six days, used daily with disposable needle tips. Excessive advertising for Saxenda has also been brought under guidelines to abide by medical ethics.
“Advertisements that mislead patients to believe Saxenda can be a guaranteed method to lose weight are deterred,” said the KMA representative. “Behavioral therapy must be paired with Saxenda for promised weight loss effects. If the taker’s lifestyle does not change, Saxenda will only give a temporary result: They will easily revert to their original weight.” Clinical trials have revealed that the Saxenda effect can last up to a year when carried out together with exercise and a healthy diet.
That Saxenda is a subcutaneous self-injectable drug is not an advantage. Patients usually prefer taking pills than having to stick themselves with needles, according to professor Kim. Keeping the pen safe for use is another matter. The pen must be kept refrigerated at all times, and when it has to be carried on a flight it must be in carry-on luggage, preferably with the doctor’s prescription. If traveling with Saxenda to a country with temperatures over 30 degrees Celsius, the injection should be kept in a flask with ice, but not in direct contact with the ice.
Saxenda should not be taken by patients with a history of medullary thyroid carcinoma, a type of thyroid cancer, or Multiple Endocrine Neoplasia syndrome type 2, an endocrine system condition.
“We are aware of the overheated attention on Saxenda in Korea as a diet injection, and see this as a problem to address,” a Novo Nordisk Korea representative told The Korea Herald.

Novo Nordisk Korea is running a campaign called “Know Saxenda Right,” which emphasizes the drug’s role as an obesity treatment that requires appropriate medical guidance. The company plans to continue and expand the campaign to include the wider public, the official said.
Globally, Novo Nordisk is running campaigns with experts to increase knowledge of obesity as a chronic disease. Novo Nordisk gives ample warning that Saxenda can cause thyroid tumors including cancer, inflammation of the pancreas, gallbladder problems, low blood sugar in type 2 diabetes patients, increased heart rate, kidney problems, allergic reactions, depression or thoughts of suicide.
“The hype right now is much to blame on irresponsible word-of-mouth reviews on social media,” Kim said, adding that a doctor would be able to advise which appetite suppressant is best for a patient depending on his or her eating habits. Whether a person’s main diet includes much rice, or beer, can be a determining factor for prescriptions for varying suppressants, Kim said.
Suppressant market
Although Saxenda is recently all the rage, holding on to the top spot for suppressants -- in the form of pill or injection -- is Ildong Pharmaceutical’s Belviq (lorcaserin), which came out in February 2015, followed by Daewoong Pharmaceutical’s Dietamin tap (pentamine), launched July 2005. Saxenda comes third. Of the three, Saxenda and Belviq have the upper hand for being able to be prescribed long-term, while Dietamin’s use is limited to three months due to the potential development of dependency. In the fourth quarter of 2018, Belviq recorded 2.3 billion won in sales and Dietamin 2.1 billion won, while Saxenda marked 5.6 billion won.
Not to be confused with diet pills, appetite suppressants for people at risk of obesity are key products for the market of obesity treatments in Korea, which was estimated at 80 billion won in 2018.
Novo Nordisk Korea imports and Zuellig Pharma Korea distributes Saxenda. Side effects experienced during pharma product usage should be reported to the Korea Institute of Drug Safety and Risk Management at 1644-6223.
By Lim Jeong-yeo (kaylalim@heraldcorp.com)









![[Hello India] Hyundai Motor vows to boost 'clean mobility' in India](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/25/20240425050672_0.jpg&u=)








