Medytox, South Korea’s largest maker of botulinum toxin, said Thursday that it has filed a lawsuit claiming intellectual property theft against domestic rival Daewoong Pharmaceutical and its US partner Alphaeon Corp.
Botulinum toxin, or BTX, is a biopharmaceutical substance widely used to cosmetically remove facial wrinkles by temporarily paralyzing muscles. The drug is based on a neurotoxin protein produced by BTX-generating bacteria.
Botulinum toxin, or BTX, is a biopharmaceutical substance widely used to cosmetically remove facial wrinkles by temporarily paralyzing muscles. The drug is based on a neurotoxin protein produced by BTX-generating bacteria.
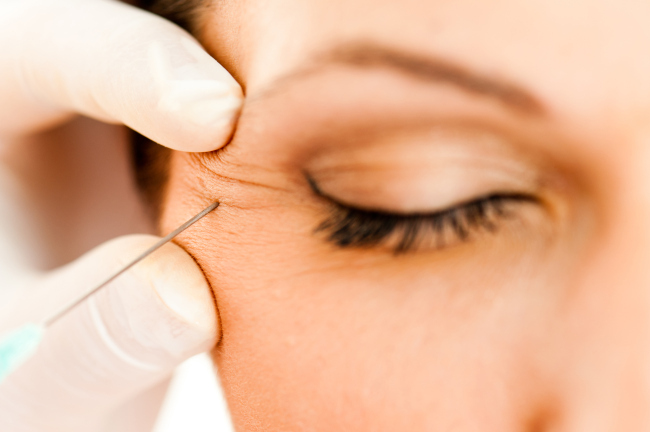
Medytox has continuously claimed that Daewoong stole its BTX bacterial strain in developing its product, setting off public controversy in Korea last year. The Seoul-based company has now taken the same issue to court in the US.
Medytox’s move comes as Daewoong and Alphaeon have filed Type A botulinum toxin Nabota for approval by the US Food and Drug Administration as of last month. If approved, Nabota is set to become the first BTX product developed by a Korean company to hit the US market.
According to court documents, Medytox on June 7 filed a lawsuit against Daewoong Pharmaceutical and Alphaeon, claiming that “DWP misappropriated Medytox’s valuable assets and related intellectual property so that DWP could avoid incurring the risk, time and expense of independently developing their own drug.”
In response to the suit, Daewoong Pharmaceutical said it was “looking into the lawsuit though it has yet to receive an official notice from the court.”
The company rejects all of Medytox’s assertions and will work to prove its innocence in the courts and to hold Medytox accountable for its actions, it said.
“Korean authorities have already ruled Daewoong innocent of all charges with regard to this case. We plan to actively address the US lawsuit and to push for strong legal actions against Medytox,” a Daewoong spokesperson said.
Medytox’s suit was filed by its legal representative Sheppard, Mullin, Richter & Hampton LLP at the Orange County Court in California, according to court documents available on the web.
Medytox claimed that a former employee named BK Lee, who was involved in the development of its BTX product Meditoxin and the Meditoxin manufacturing production line, breached the firm’s confidentiality agreement.
The company argues that Lee stole Medytox’s BTX strain and trade secrets on producing its botulinum toxin brand Meditoxinm, and delivered the information to executives at Daewoong Pharma in exchange for financial rewards.
It also claims that Daewoong paid BK Lee more than $120,000 and provided favors to the former employee, including paid positions during Lee’s post-doctorate studies at Purdue University, in exchange for Medytox’s trade secrets.
As news of the lawsuit was publicized Thursday, Daewoong Pharmaceutical shares plunged 7 percent to a close of 93,000 won ($83) on the day. Meanwhile, Medytox shares gained 1.19 percent to a close of 595,000 won.
In addition to Daewoong, other Korean drugmakers including Medytox and Hugel are looking to launch their own BTX products in the US in the near future.
Currently, the US botulinum toxin market, worth $1.5 billion, is dominated by Allergan with an 85 percent market share.
Hugel is currently carrying out phase 3 clinical trials for its own BTX Botulax with aims to obtain the US FDA’s approval by next year.
There has been speculation that Medytox, which has partnered with Allergan, will begin phase 3 clinical trials for its liquid-injectable BTX Innotox by the end of this year.
By Sohn Ji-young (jys@heraldcorp.com)





![[From the Scene] Monks, Buddhists hail return of remains of Buddhas](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/19/20240419050617_0.jpg&u=20240419175937)





![[Graphic News] French bulldog most popular breed in US, Maltese most popular in Korea](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/18/20240418050864_0.gif&u=)


![[From the Scene] Monks, Buddhists hail return of remains of Buddhas](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=652&simg=/content/image/2024/04/19/20240419050617_0.jpg&u=20240419175937)

![[KH Explains] Hyundai's full hybrid edge to pay off amid slow transition to pure EVs](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=652&simg=/content/image/2024/04/18/20240418050645_0.jpg&u=20240419100350)

![[Today’s K-pop] Illit drops debut single remix](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=642&simg=/content/image/2024/04/19/20240419050612_0.jpg&u=)